| Abstract No.: | C-F3178 |
| Country: | Canada |
| | |
| Title: | DEVELOPMENT OF A LIPOSOMAL FORMULATION FOR THE TREATMENT OF PRIMARY BRAIN TUMORS |
| | |
| Authors/Affiliations: | 1 Marc-André Bellavance*; 1 Fernand Gobeil; 1 David Fortin;
1 Sherbrooke University, QC, Canada
|
| | |
| Content: | Objectives: Systemic treatment of many central nervous system (CNS) diseases, such as brain tumors, is considerably impaired by limited delivery of therapeutics to the CNS by an intrinsic obstacle, the blood-brain barrier (BBB). Unfortunately, very few molecules can cross the intact BBB and even for the ones who do, CNS entry is not assured. In fact, highly active efflux pumps expel these molecules back in the bloodstream. As a consequence, 98% of small molecules and the totality of large molecular weight drugs are excluded from the CNS compartment.
Therefore, different strategies have been developed to improve CNS delivery. One of these strategies involves the use of an inert vehicle such as liposomes to encase the therapeutic solution. We undertook the design of a custom made liposome formulation that would possess the features allowing its entry across the BBB.
Material and Methods: After a thorough review, we selected the following lipids for a first liposomal formulation: DSPC (65%), ovine wool cholesterol (30%) (Avanti Polar Lipids, USA) and rhodamine DHPE (Invitrogen, CA) . This lipid mixture was dissolved in 2 mL of chloroform and a thin lipid film was obtained by removal of the solvent by rotary evaporation at 65°C, 130 rpm during 1 hour. The lipid film was then hydrated with 2 mL of a 20mM HEPES buffer pH 7,4 containing 150 mM NaCl and 5 mM of 5-carboxyfluorescein (Calbiochem, CA). The resulting lipid suspension was shaken during 1 hour at 65°C and frozen and thawed 5 times (liquid nitrogen/water bath at 65°C). Liposomes were extruded 10 times through 2 stacked polycarbonate membranes with a pore size of 100 nm with a Lipex Extruder (Northern Lipids, CA). Liposomes were purified by chromatography with a pre-packed PD-10 Sephadex G-25 M column (Bio-Rad, CA). The final suspension was stored in glass vials at 4°C.
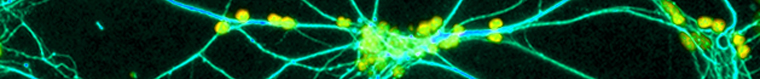
Size and thermal stability of the liposomes were determined by transmission electron microscopy. Lipofection of F98 cells was conducted by adding 50, 100 and 200 μl of the final liposomes suspension to 1 mL of supplemented DMEM (Wisent, CA) and incubating with cells for 10hrs, at 37°C and 5% CO2. Delivery efficacy was quantified by FACS analysis. Confocal microscopy was also employed to monitor the localization of the fluorophores in the transfected cells.
Results: Narrow distribution of liposomes of 80 to 140 nm were obtained with the method described herein. The optimal storage temperature was 4°C; liposomes preserved their size and shape for at least 96 hrs under this temperature. The maximal F98 lipofection measured by FACS was 93%. Confocal microscopy revealed that both liposomal markers co-localized in cytosol of cells, therefore suggesting endocytosis as the delivery mechanism.
Conclusion: These promising results show that high lipofection levels of F98 can be obtained with this liposomal formulation in vitro. In vivo experiments are under way to determine if the liposomes are successful to circumvent the BBB and access the brain parenchyma. |
| | |
| Back |
|